STUDYING CANCER STEM CELLS
Overview
Cancer stem cells (CSCs) are thought to be a major driver behind the aggressiveness of GBM. Essentially, CSCs are a subgroup of cells that are present within the tumor. Like normal stem cells, they have the ability to differentiate to every cell type required in the tumor and they have the ability to continually self-renew. In addition, they display a tremendous ability to respond to environment stressors, including chemotherapy and radiation. As such, when tumors are exposed to chemotherapy, although most cells are killed, CSCs will often survive and will invade into normal tissue. Over time, they are then able to divide and differentiate in order to eventually recapitulate the entire tumor (Figure 1). This is thought to be one the main reasons behind the resistance and recurrence that is seen in GBM. (For more information about GBM resistance and recurrence - see here). Given that they are such an important driver, studies around them are focused on understanding their properties and understanding mechanisms they use to be so resistant so that we can target them more effectively (1).
Figure 1: CSCs in GBM
Approaches
There are many different ways to approach understanding CSCs. Early studies were more focused on understanding how to identify these cells and determine that they were truly stem cells (1). From this, we were able to identify CD133 and CD15 as reasonable GBM stem cell markers. We were also able to understand that neurosphere assays are a good way to look at stemness potential. More recent studies have focused on understanding specific mechanisms and pathways that are upregulated in these stem cells, and that are therefore likely allowing them to be so aggressive (1). Even more recently, novel therapeutic strategies have been tried that specifically target stem cells. Unfortunately, we have yet to identify a specific strategy that is effective, but there is still much research to be done in this area.
Our Work
Our lab has worked on identifying genes and mechanisms that may be important for maintenance of this cancer stem cell niche. Some examples of our projects include working on understanding how GSCs develop over time after exposure to therapy. We have also looked at the role of HIF signaling and the role of that signaling in GSC expansion and development. A few examples of our work are below - see more by looking at the entire publication list.
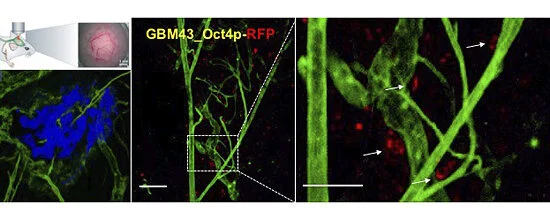
CSCs were tagged using a Oct4-RFP reporter construct. Here, we see them in our in vivo tumor model, localizing near blood vessels.
Therapeutic stress can induce conversion to a stem cell state, which can then lead to recurrence of the tumor.